Expiration date: 01/2026
Convenia
for the treatment of diseases of bacterial etiology in dogs and cats
(organization-developer, the company "Zoetis Inc", USA)
I. General information
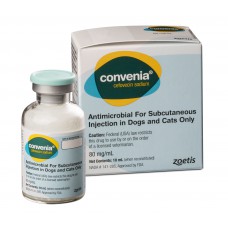
Trade name of the drug: Convenia (Convenia).
International nonproprietary name: cefovecin.
Dosage form: lyophilized powder for preparation of solution for injection.
Convenia as an active substance contains cefovecin (in the form of sodium salt) and is available in two packs complete with solvent (in a separate bottle):
Convenia in a bottle with a capacity of 20 ml contains cefovecin-852 mg and excipients: methyl parahydroxybenzoate-19.17 mg, propyl parahydroxybenzoate-2.13 mg. the Solvent contains benzyl alcohol-13 mg / ml and water for injection-10.8 ml.
Convenia in a bottle with a capacity of 5 ml contains cefovecin-340 mg, as auxiliary substances: methyl parahydroxybenzoate-7.67 mg, propyl parahydroxybenzoate-0.85 mg. the Solvent contains benzyl alcohol-13 mg / ml and water for injection-4.45 ml.
In appearance, the Convenia is a lyophilized powder from white to yellow, solvent – transparent, colorless liquid.
The Convenia is produced sterile packaged in glass bottles with a capacity of 5 ml and 20 ml, the solvent – in glass bottles with a capacity of 10 ml and 15 ml, respectively, sealed with rubber stoppers, reinforced with aluminum caps. Bottles of appropriate volumes, one with and one lyophilisate with solvent Packed in a cardboard carton together with instructions for use.
Store the drug in a closed package of the manufacturer, in a dry, dark place, away from food and feed at a temperature of 2°C to 8°C.
Shelf life of the drug under storage conditions is 3 years from the date of production, shelf life of the prepared therapeutic solution at a storage temperature of 2°C-8°C — 28 days.
It is forbidden to use the drug after the expiration date.
The Convention should be kept out of the reach of children.
Unused product is disposed of in accordance with the law.
II. Pharmacological properties
Convenia relates to antibacterial drug of the cephalosporin group III generation.
Cefovecin – active substance Convenia has a broad spectrum antibacterial activity against most gram-positive and gram-negative microorganisms, including Staphylococcus pseudintermedius and Pasteurella multocida, are usually released during infections of the skin in dogs and cats; Prevotella oralis, Bacteroides spp. and Fusobacterium spp., secreted from abscesses in cats; Porphyromonas spp. and Prevotella spp, secreted in acute infectious and inflammatory periodontal and gum diseases in dogs, and Escherichia coli, secreted in urinary tract infections in dogs and cats.
The mechanism of action of cefovecin is to block the transpeptidase enzymes that bind the peptidoglycan of the cell wall, resulting in the bacterial cell is destroyed.
Cefovecin is rapidly absorbed from the injection site, reaching a peak concentration in blood plasma in dogs after 6 hours, in cats-after 2 hours. Therapeutic concentrations of the antibiotic after a single administration of the drug in the body of dogs and cats persist for 14 days.
Cefovecin more than 96% bound to plasma proteins, excreted mainly in the urine unchanged, the half-life of the drug in dogs-5.5 days, cats-6,9 days.
According to the degree of impact on the body of the Convention refers to the" low-risk substances " (4 hazard class according to GOST 12.1.007-76).
III. The order of application
Convenia prescribed to dogs and cats for the treatment of diseases of the skin and soft tissues (abscesses, pyoderma) and urinary tract infections, bacterial etiology, caused by sensitive to the action of cefovecin microorganisms. For acute infectious-inflammatory diseases of the gums and periodontal tissues associated with Porphyromonas spp. and Prevotella spp., Convenia used dogs in the complex treatment (with surgical treatment).
It is forbidden to use the drug to dogs and cats with hypersensitivity to (beta-lactam antibiotics, pregnant females, puppies and kittens up to 8 weeks of age, as well as animals during mating.
Convenia used dogs and cats in the form of solution for injection, which is prepared before use, in compliance with the rules of asepsis. In the bottle with the antibiotic powder, add the attached solvent and shake thoroughly until it is completely dissolved.
The prepared therapeutic solution containing 80 mg of cefovecin in 1 ml is administered once subcutaneously at a dose of 1 ml per 10 kg of animal weight (equivalent to 8 mg of cefovecin per 1 kg of animal weight).
If necessary, the drug is administered again in the same dose at intervals of 14 days. The total course of treatment should not exceed four injections.
Symptoms of drug overdose were not revealed.
Features of the drug at its first application and cancellation is not established.
If necessary, re-administration of the drug should be avoided violations of the recommended interval between injections, as this can lead to a decrease in therapeutic efficacy. In case of missing another injection, the drug should be administered as soon as possible in the same dose.
When using the drug in accordance with this instruction, side effects and complications, as a rule, are not noted. In the case of allergic reactions, the use of the drug is stopped and antihistamines and symptomatic therapy are prescribed.
It should not be used in conjunction with Konveniyu furosemidom, ketoconazole, nonsteroidal anti-inflammatory drugs and other drugs that have a high level of plasma protein binding, in connection with the possible occurrence of side effects.
The drug is not intended for use in productive animals.
IV. Personal prevention measures
When working with the Convention should follow the General rules of personal hygiene and safety provided when working with drugs for veterinary use. During work it is forbidden to drink, smoke, eat, at the end of the work it is necessary to wash your hands thoroughly with soap.
In case of accidental contact of the drug with the skin or mucous membranes, they should be washed with plenty of water.
People with hypersensitivity to beta-lactam antibiotics should avoid direct contact with the Convention. In case of allergic reactions or accidental ingestion of the drug into the human body, you should immediately contact a medical institution (you should have instructions for use of the drug or a label).
Empty packaging of the drug shall not be used for domestic purposes, it shall be disposed of with household waste.
Organization-manufacturer: "Haupt Pharma Latina S. r.l.", Borgo san Michele (LT) ss 156km50 - Latina, Italy.
The manual was developed by " Zoetis Inc."(100 Campus Drive , Florham Park, New Jersey, 07932 USA), together with Zoetis (123317, Moscow, Presnenskaya embankment, 10.)